 I assembled my blot sandwich according to instructions: on the bottom, a stack of transfer pads (which comprises of 7 layers of filter pads), followed by my nitrocellulose sheet, followed by my gel, followed by another transfer stack. trans-blot turbo mini size nitrocellulose trans-blot turbo mini size transfer stacks Bio rad 5x transfer buffer (when I dilute it, I put ethanol, following their instructions, and water) 4~20% SDS-PAGE Gels (I also used one made by myself. I'm using Bio-Rad equipment and reagents, being them: 
My gel gets cleaner as it can be, and my membrane gets nothing. I get to see all the bands from my molecular weight standard on the gel, everything is just fine, but when I put it to transfer on a semi-dry apparatus, they just fucking vanish. 1, ,2), 2), a process normally carried out in a period of up to 2 days.I need to do a western blot (fuck this assay) of two proteins (namely Gal-3 and TLR-4), and I'm having an awful ride when transfering them.ĭuring electrophoresis, everything runs smoothly. Here we show that the entire process of electrophoresis of autoantigens Ro60 and La (actual gel running), western transfer and immunoblotting with specific autoantibodies can be carried out in one hour ( Fig. Traditional immunoblotting normally takes about four to five hours, with 1 hour for blocking, 2 hours for incubation with primary antibody, 1 hour with secondary antibody, 30 min for washing between primary and secondary antibody incubation and finally development with substrate. We have shown that both low and high molecular weight proteins can be transferred very efficiently to nitrocellulose membranes in a very short time using heated transfer buffer without methanol ( 10, 11). Proteins of varying sizes (10 −250 kD) can be transferred to membranes in 3 to 7 minutes very efficiently ( 9). 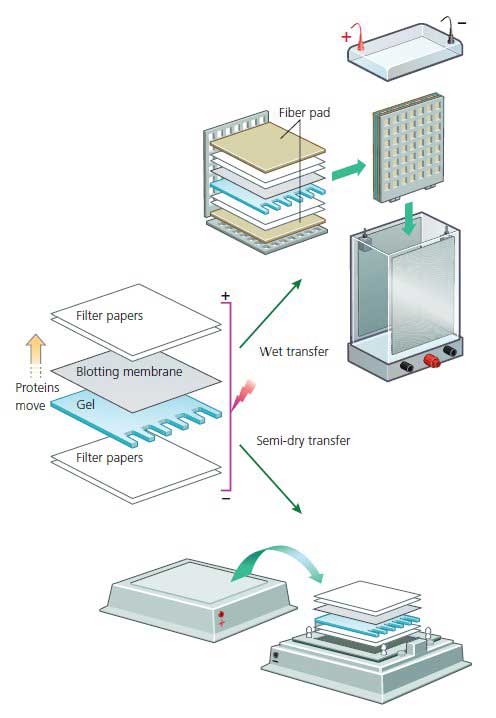
Trans-blot turbo system employs a semi-dry method of transfer of proteins from gel to membranes. Prolonged electrotransfer (16–20 h) at high current density coupled with inclusion of 0.01% sodium dodecyl sulfate, to enhance protein elution, has been used to efficiently transfer high-molecular weight proteins ( 9, 10). High molecular weight proteins are often stubbornly resistant to transfer ( 7) in spite of prolonged runs and this problem is accentuated when higher percentage gels are used. The protein transfer procedure normally takes about 2–4 hours at about 70 volts or an overnight transfer at 30 volts. Electrophoretic transfer of proteins, resolved by SDS-PAGE, to nitrocellulose is a fundamental step prior to detection of specific proteins with specific antibodies ( 7– 9). The transfer to membranes has been achieved by (a) simple diffusion ( 4) vacuum-assisted solvent flow ( 5) and (c) electrophoretic elution ( 6). However, Haeberle demonstrated with a special gel and a special buffer that was heated to 70 ˚C, that it is possible to electrophorese proteins in 5 minutes ( 3).įor immunoblotting, the separated proteins are transferred to nitrocellulose or polyvinylidene difluoride membranes. Electrophoresis of proteins on a mini-gel takes at least 2 hours to complete to ensure ideal separation without “smiling” artifacts.  
Proteins separated on SDS PAGE can visualized by either staining with various protein stains or by immunoblotting. Proteins are first separated on the basis of size in sodium dodecyl sulfate polyacrylamide gel electrophoresis (SDS-PAGE) ( 1, 2). 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
